DNA Rna Test Kit Inactivation Nasal Transport Medium Vtm Disposable Specimen Collection Virus Sampling Tube
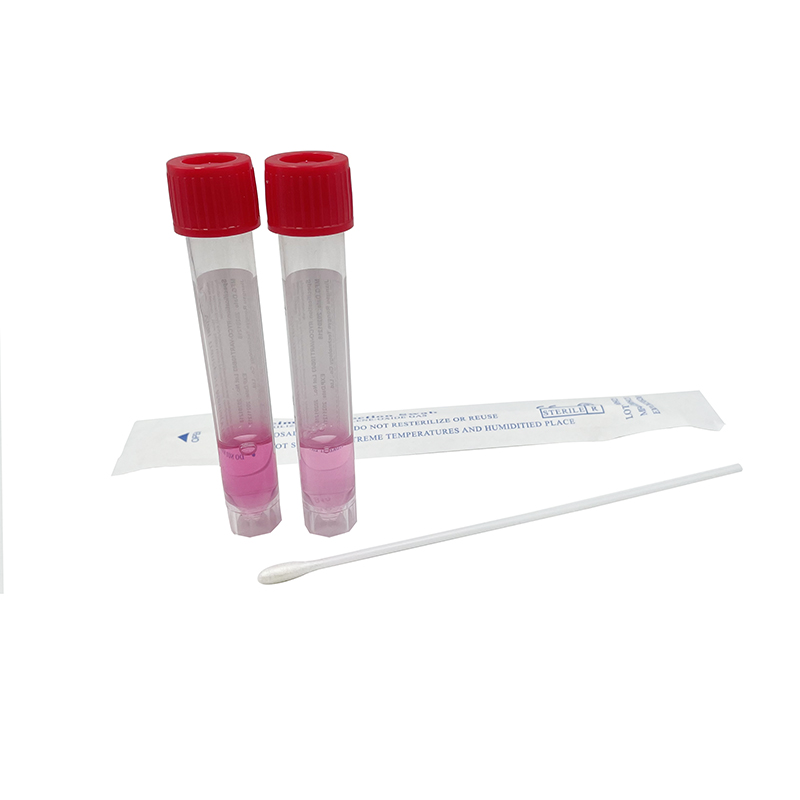
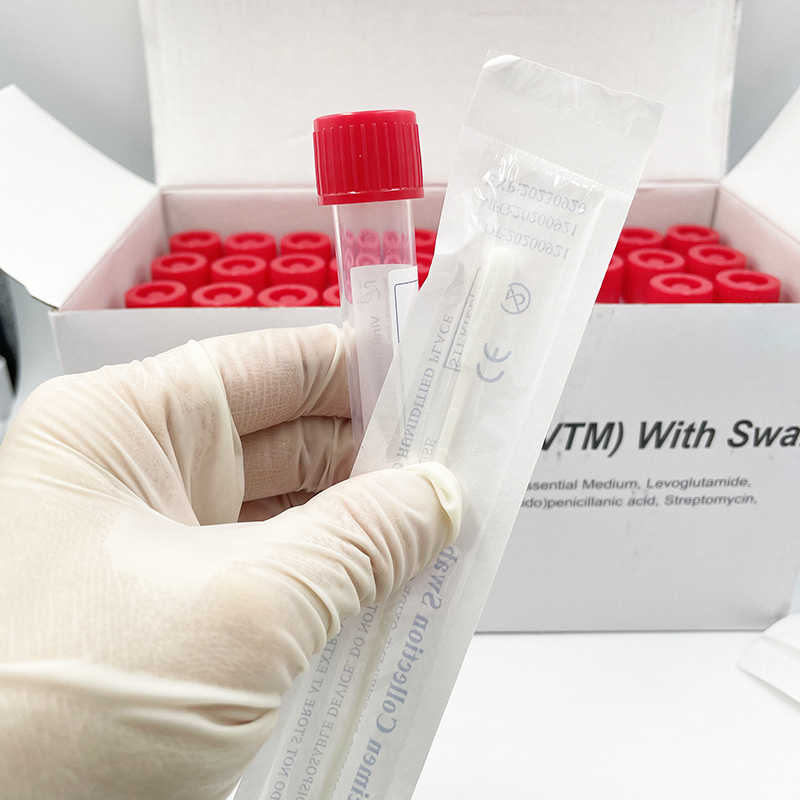
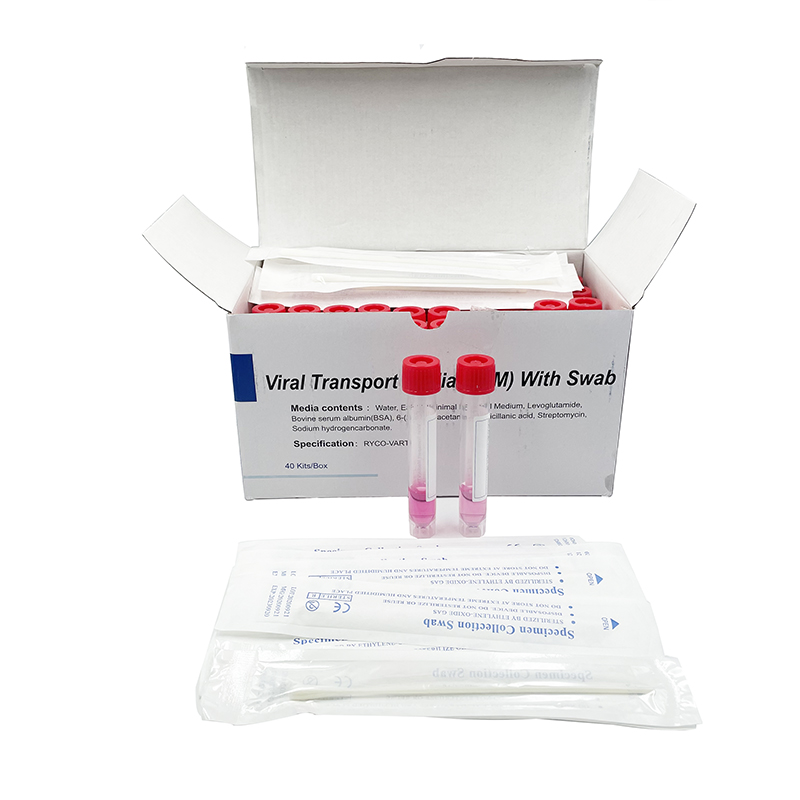
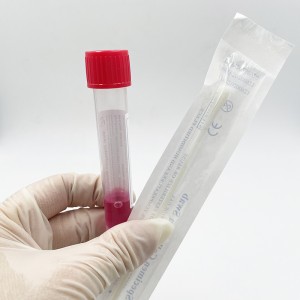
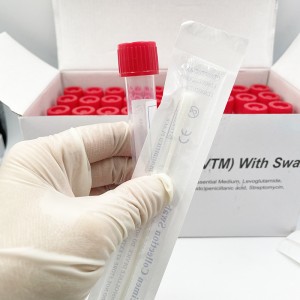
ʻO ka mea lawe maʻi viral me nā swabs
Hoʻohana ʻia ia no ka ʻohi ʻana i nā mea huna mai ka ʻāʻī a i ʻole ka ihu. ʻO nā laʻana i hōʻiliʻili ʻia e nā swabs mālama ʻia i loko o ke ʻano preservative i hoʻohana ʻia no ka hoʻāʻo ʻana i ka maʻi virus, kanu ʻana, kaʻawale a pēlā aku.
He swab naseopharyngeal ka swab, ua hoʻopili paʻa ʻia lākou, EO-sterilized, nylon flocked, 155mm me 80 mm breakpoint, CE-marked, i hana ʻia e kahi mea hana i kākau inoa ʻia e FDA, a loaʻa iā ia kahi ola 2-makahiki.

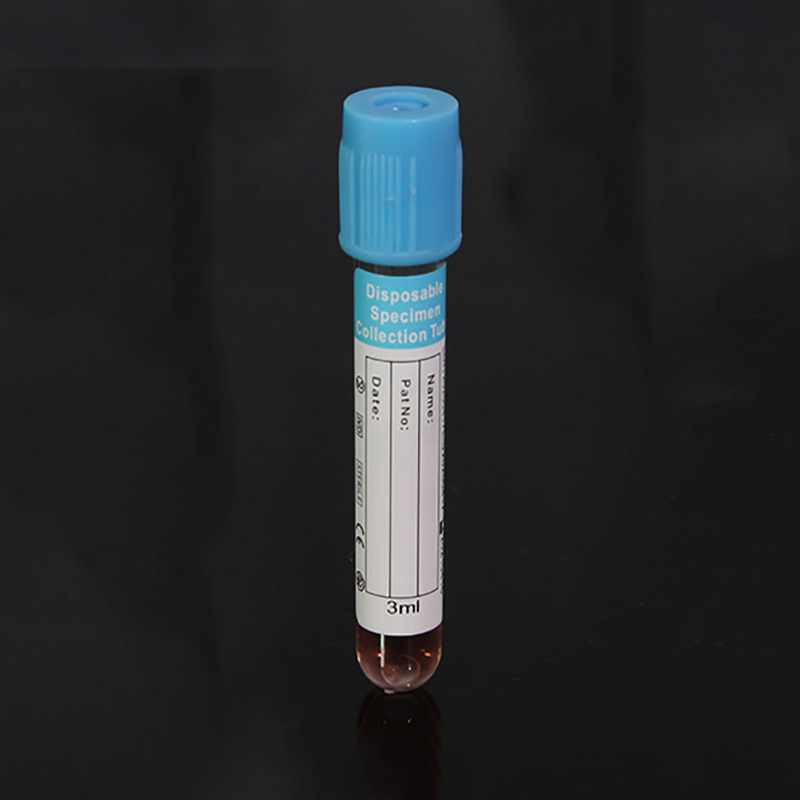
ʻO ka kūleʻa o ka maʻi maʻi o SARS-CoV-2 (2019-nCoV) i ka wā o ka maʻi COVID-19 e hilinaʻi nui ʻia i ka maikaʻi o ka specimen a me nā kūlana i lawe ʻia a mālama ʻia ka specimen ma mua o ka hana ʻia ʻana i loko o ka hale hana. Aia kekahi paipu 12 ml me 3 ml o VTM (Virus Transport Media) a me kahi swab sterile. Ua mākaukau ka media transport virus e hoʻohana a me kekahi o nā mea palekana a puni. Hoʻolālā ʻia ka media transport virus e lawe i nā maʻi, me ka coronavirus, no ka noiʻi a me ka hoʻāʻo ʻana. Hana ʻia kēlā me kēia ʻāpana o ka VTM ma lalo o nā alakaʻi koʻikoʻi e like me ka mea i kuhikuhi ʻia e ka CDC, he sterile, a loaʻa ka mana maikaʻi ma mua o ka hoʻokuʻu ʻana (E nānā i ka CoA). Paʻa ma ka liʻiliʻi ʻeono mahina ma ka lumi wela (2-40°C). Paʻa a hiki i hoʻokahi makahiki ke mālama ʻia 2-8 ° C. Loaʻa pū kekahi koho me nā ʻeke biohazard.
| Inoa | ka mea lawe viral me ka swabs |
| Volume | 1ml |
| ʻAnoʻ | ʻAʻole hana ʻole |
| Pūʻolo | 1 pahu / ʻeke pepa-pepa 40 kits / pahu 400 kits / pahu pahu |
| Palapala | CE ISO |
ISO13485
CE
EN ISO 13485: 2016/AC: 2016 ʻōnaehana hoʻokele maikaʻi o ka lāʻau lapaʻau no nā koi hoʻoponopono.
EN ISO 14971: 2012 Nā mea lapaʻau - Ka hoʻohana ʻana i ka hoʻokele pilikia i nā mea lapaʻau
ISO 11135: 2014 Mea lapaʻau Sterilization o ka ethylene oxide Hōʻoia a me ka mana maʻamau
ISO 6009: 2016 Nā nila hoʻoheheʻe sterile hoʻokuʻu e ʻike i ke code kala
ISO 7864: 2016 ʻO nā nila hoʻoheheʻe sterile hoʻopau
ISO 9626: 2016 ʻO nā paipu nila kila kila no ka hana ʻana i nā mea lapaʻau

He alakaʻi nui ʻo SHANGHAI TEAMSTAND CORPORATION i nā huahana lapaʻau a me nā hoʻonā.
Me ka ʻoi aku o 10 mau makahiki o ka ʻike hoʻolako olakino, hāʻawi mākou i kahi koho huahana ākea, nā kumu kūʻai hoʻokūkū, nā lawelawe OEM kūʻokoʻa, a me ka hāʻawi ʻana i ka manawa kūpono. ʻO mākou ka mea hoʻolako o ka Australian Government Department of Health (AGDH) a me California Department of Public Health (CDPH). Ma Kina, kū mākou i waena o nā mea hoʻolako kiʻekiʻe o Infusion, Injection, Vascular Access, Rehabilitation Equipment, Hemodialysis, Biopsy Needle a me nā huahana Paracentesis.
Ma ka 2023, ua hoʻopuka maikaʻi mākou i nā huahana i nā mea kūʻai aku ma 120+ mau ʻāina, me ka USA, EU, Middle East, a me Asia Hikina Hema. Hōʻike kā mākou mau hana i kēlā me kēia lā i ko mākou hoʻolaʻa ʻana a me ka pane ʻana i nā pono o ka mea kūʻai aku, e hoʻolilo iā mākou i hoa ʻoihana hilinaʻi a hoʻohui ʻia i koho.

Ua loaʻa iā mākou ka inoa maikaʻi ma waena o kēia mau mea kūʻai aku no ka lawelawe maikaʻi a me ke kumukūʻai hoʻokūkū.

A1: Loaʻa iā mākou he 10 mau makahiki ma kēia kahua, ʻO kā mākou hui he hui ʻoihana a me ka laina hana ʻoihana.
A2. ʻO kā mākou huahana me ke kūlana kiʻekiʻe a me ke kumukūʻai hoʻokūkū.
ʻO A3.ʻO ka maʻamau ka 10000pcs; makemake mākou e hui pū me ʻoe, ʻaʻohe hopohopo e pili ana i ka MOQ, e hoʻouna wale mai iā mākou i kāu mau mea āu e makemake ai e kauoha.
A4.Yes, ʻae ʻia ka hoʻopilikino LOGO.
A5: Ke mālama nei mākou i ka hapa nui o nā huahana i ka waihona, hiki iā mākou ke hoʻouna i nā laʻana ma 5-10workdays.
A6: Hoʻouna mākou e FEDEX.UPS, DHL, EMS a i ʻole Sea.